button at the top of the page.
button at the top of the page.The Clinical Trial Application Menu is the hub of the EudraCTEudraCT (European Union Drug Regulating Authorities Clinical Trials) is the European Clinical Trials Database of all interventional clinical trials of medicinal products commencing in the European Union from 1 May 2004 onwards. The EudraCT database has been established in accordance with Directive 2001/20/EC. system:
 button at the top of the page.
button at the top of the page.
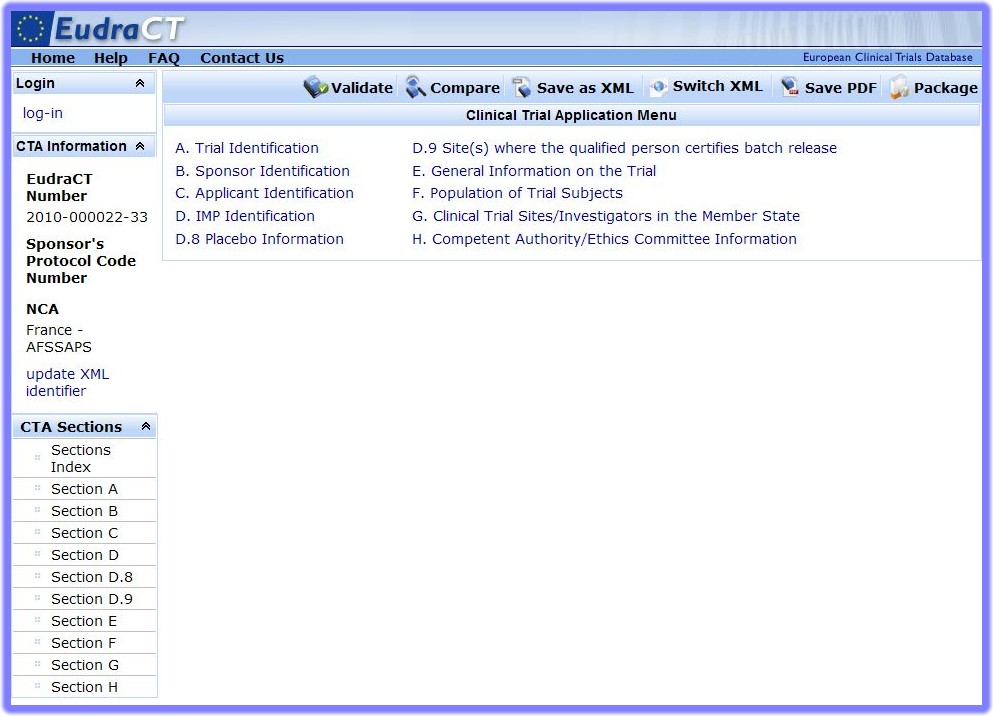
From this starting point, users can make a Clinical Trial Application within the EEAAcronym: European Economic Area. Created in 1994, the EEA combines the countries of the European Union and member countries of EFTA (European Trade Association) Countries that belong to the EEA are: Austria, Belgium, Bulgaria, Czech Republic, Cyprus, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Latvia, Liechtenstein, Lithuania, Luxembourg, Malta, Netherlands, Norway, Poland, Portugal, Romania, Slovakia, Slovenia, Spain, Sweden, United Kingdom. Countries that are EEA member countries but NOT part of the European Union are: Norway, Iceland, and Liechtenstein., or in a Third CountryA third country is a country outside of the European Union/European Economic Area.. This completed, a package can be created, ready for submission to a National Competent Authority or to the EMAAcronym: European Medicines Agency.
In addition, registered users, such as PIPAcronym: Paediatric Investigation Plan. Document upon which the development and authorisation of medicinal products for the paediatric population is based. It is presented by an applicant early during the development of a product to the EMA Paediatric Committee in order to agree a paediatric development plan. addresses can log in to the system to perform tasks relating to their roles.
XMLAcronym: eXtensible Markup Language documents of the CTAAcronym: Clinical Trial Applications may be uploaded from a local area using the  Clinical Trial Application button. "Load a Clinical Trial Application or Third Country CT Information".
Clinical Trial Application button. "Load a Clinical Trial Application or Third Country CT Information".
 in the Top bar:
in the Top bar: